
To help you confidently consult with patients who may be eligible for natural therapy, we’ve put together a list of the top five questions prescribers typically ask about alternative medicine.
If you’re already prescribing natural therapeutics to patients or you are about to write your first natural therapeutic script, read this blog to help you on your journey.
Is my patient eligible for natural therapy treatments?
In Australia, a medical practitioner can apply to access natural therapeutics for a patient if they believe it is clinically suitable.
Although there is no set criteria or list of conditions, there are two general situations that will typically make your patient an ideal candidate, including:
- Patient has a chronic medical condition that has lasted longer than three months – The TGA has approved alternative medicine as a second-line treatment for a number of medical conditions. There is no official list of pre-approved conditions from the Therapeutic Goods Administration (TGA) for prescribing doctors to reference as applications can be made for any patient if you think it is clinically suitable.
- Patient has not responded positively to conventional medical treatment including over the counter or prescription medications – If conventional or first-line medication did not relieve the symptoms of your patient’s condition or improve their quality of life, or if the medication caused serious or intolerable side effects, they may be eligible.
Before prescribing, a few other questions you should ask yourself are:
- Have I reviewed evidence for natural therapeutic use for the patient’s condition?
- Is natural therapy appropriate for this patient?
- Is there potential for drug interactions? P450 metabolisers?
- Will there be an increased need for therapeutic monitoring?
- Do I have the relevant expertise to prescribe natural therapy?
- Do I know enough about the product I am prescribing?
What drug schedule is natural therapy classified as?

In Australia, most alternative medicines are still currently unapproved therapeutic goods. This means the TGA has not assessed them for safety, quality or effectiveness and they are therefore not registered on the Australian Register of Therapeutic Goods (ARTG).
Alternative medicine products are currently classified as either Schedule 3, Schedule 4 or Schedule 8 substances under the Poisons Standard set out by the TGA.
- Schedule 3 (Pharmacist Only Medicine) – This applies to any low-dose CBD (cannabidiol) products containing up to a maximum of 150 mg/day of CBD. Please note, there are currently no Schedule 3 CBD products available in Australia as they must first be approved by the TGA and included on the Australian Register of Therapeutic Goods (ARTG) to be considered an S3 substance.
- Schedule 4 (Prescription Only Medicine) – This applies to any natural therapeutic that contains less than 2% THC (tetrahydrocannabinol). So in other words, the product must have more than 98% CBD content to be considered a schedule 4 substance.
- Schedule 8 (Controlled Drug) – This applies to any alternative medicine product that contains more than 2% THC content. As most natural therapeutics currently on the market contain a variation of CBD and THC quantities (higher than 2% THC), most natural therapeutics are classified as Schedule 8 substances.
As most alternative medicine products are not registered on the ARTG, prescribing doctors must first apply to the TGA for approval to access these particular products for their patients with a prescription.
What’s the difference between the Special Access Scheme (SAS) and Authorised Prescriber Scheme access pathways?

If you determine that natural therapy is clinically suitable for your patient, you will need to apply through the TGA’s Special Access Scheme (SAS) or Authorised Prescriber Scheme before you provide your patient with the prescription.
Although there are a few different avenues within these pathways that doctors can take, the most commonly utilised ones that doctors apply through are the Authorised Prescriber and the Special Access Scheme Category B (SAS-B) application pathway.
- Authorised Prescriber pathway – Any Australian-registered medical practitioner can apply to become an Authorised Prescriber of certain natural therapeutics. Within this pathway, doctors can apply through either the ‘Standard pathway’ or the ‘Established History of Use pathway’. Unlike the standard pathway, the Established History of Use pathway does not require approval from a Human Research Ethics Committee or endorsement from a specialist college. Doctors prescribing through this pathway can choose products from a list of medicines with established history of use.
- SAS Category B application pathway – Any prescribing healthcare practitioner including medical practitioners and nurse practitioners can apply to the TGA to access natural therapeutics for an individual patient using this pathway. Applications will be assessed by the TGA on a case-by-case basis. Prescribers applying through this pathway must provide a clinical justification summarising the following:
- patient’s symptoms and/or diagnosis
- details of relevant past treatments and procedures trialled or considered, plus reasons why therapeutic goods on the ARTG may not be an appropriate treatment for the individual patient
- an assessment of the expected clinical benefits versus the potential risks of the proposed treatment.
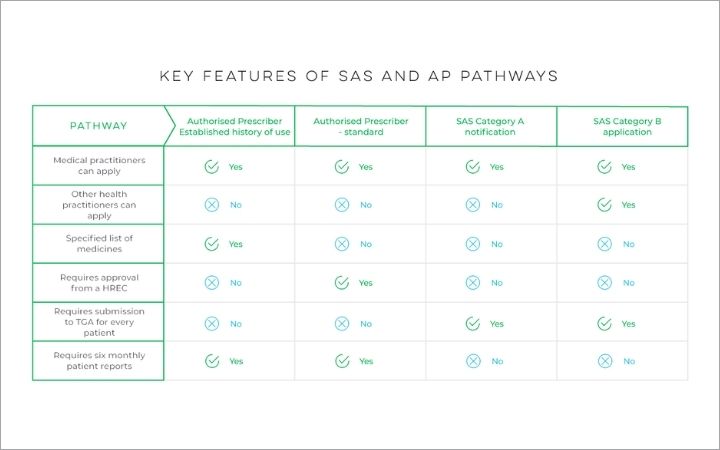
Information retrieved on 23/05/2022 from TGA Key features of SAS and AP pathway table.
How do I dose natural therapeutics
Another common question asked by prescribers is how to dose natural therapeutics. As natural therapy is still classified as an unapproved treatment option, there are no pre-defined dosage requirements or set dosing schedules that prescribing doctors must follow.
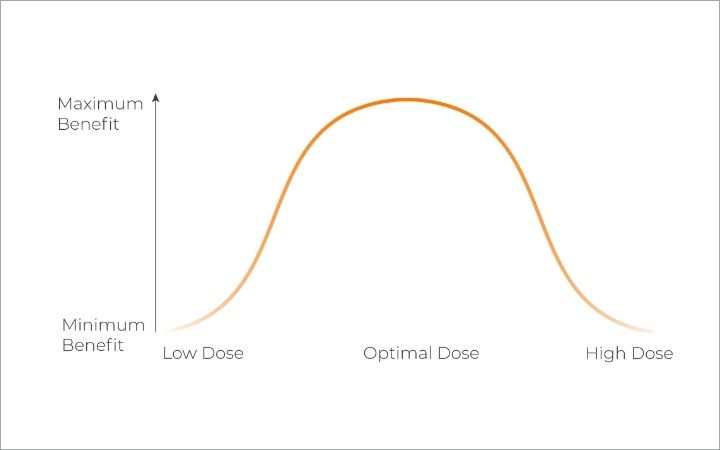
To find the right dose for your patient, it is recommended to follow the ‘start low and go slow’ approach.
This approach requires the doctor to utilise the titration dosing method, where you can monitor the patient’s treatment response to identify safe and effective dosing at individualised levels.
By harnessing the self-titration dosing method, you can work with your patient to help them find the most suitable dose where the desired therapeutic effect is achieved.
One important learning to understand is that natural therapeutic compounds have biphasic properties. This means that low and high doses can produce opposite effects. Small doses have commonly been found to stimulate while large doses tend to sedate.
Just like any medicine, natural therapeutics may not work for everyone. So it’s important to monitor your patient’s responses throughout their treatment plan and adjust the dosing accordingly.
For more information, you can read more in our blog on finding the right dose for your patient.
How do I find alternative medicine products available to prescribe?
Currently in Australia, there are over 350 natural therapy products on the market which are available to prescribe. You can either go directly to the wholesaler to find information about each individual brand and product, or you can use a platform such as Canview to explore multiple products in one place.
Canview platform provides Australian doctors with access to the widest range of natural therapeutics in the country.
With more than 350 products available, medical practitioners can register on Canview to view and compare products, download CMI information and view each products’ Certificate of Analysis.
With all the information in one place, you can use this platform to make informed decisions before and during your patient consults to find the most suitable treatment option for each patient’s circumstances.
On Canview, you can also download clinical resources and take our free Confident Prescriber Course, which will give you the knowledge and tools to confidently consult with patients and prescribe natural therapeutics.

References:
Disclaimer: The contents in this article do not constitute legal advice, are not intended to be a substitute for legal advice and should not be relied upon as such. You should seek legal advice or other professional advice in relation to any matters you or your organisation may have.
Canview does not endorse the use of natural therapeutics without lawful prescription. Just like any medicine, natural therapeutics may have both positive and negative side effects on the user and should only be prescribed to patients by a health professional with the authority and expertise to do so. The information provided by Canview is for informational and educational purposes and is of a general nature. Patients considering natural therapy are advised to speak to their general practitioner first to see if it’s a suitable therapy.