Reshaping how Australians access alternative medicine
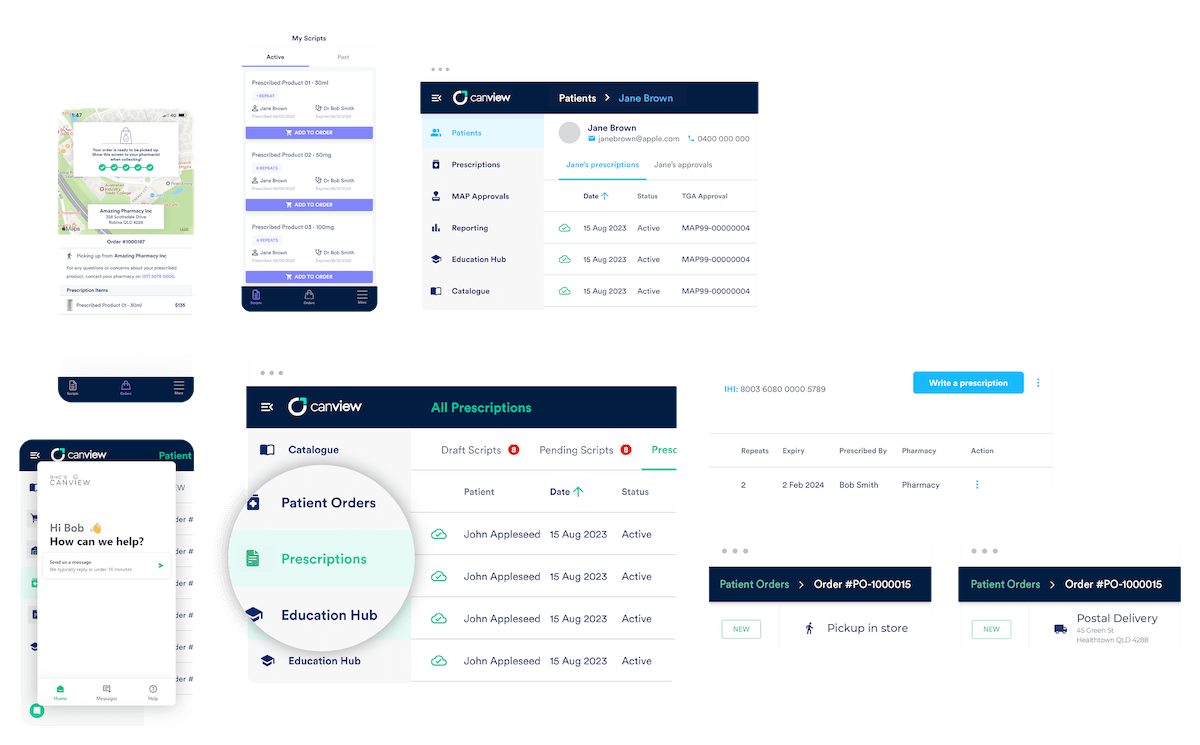
An intuitive software backed by simple integrations and machine learning, Canview eliminates the hassle previously associated with prescribing and ordering alternative medicine, such as plant-based medicine and smoking cessation products. Not only is Canview a single destination to access Australia’s widest range of controlled plant-based therapies, it also delivers:
- Seamless prescribing and approval management
- Innovative dispensing and order management
- Transparent prescription management for patients
Join thousands of healthcare professionals and transform the way you prescribe, order and dispense with Canview.

Building confidence through education
In an ever evolving and emerging industry, navigating the landscape of emerging therapeutics can be difficult – even for seasoned professionals. Canview provides education that will elevate your prescribing or dispensing confidence.
Register to gain access to our expanding Education Hub, designed by health professionals, for health professionals. Register to browse:
- Canview Confident Prescriber Course
- Canview Confident Dispensing Course
- Smoking cessation education
- Links to CPD accredited webinars
- Downloadable clinical resources